It’s Time to Evolve Beyond FMTs and Advance LBPs
The FMT era has been productive, but now it’s time to rally together as an industry to accelerate next-generation LBP drug discovery, development and FDA approval.

The FMT era has been productive, but now it’s time to rally together as an industry to accelerate next-generation LBP drug discovery, development and FDA approval.
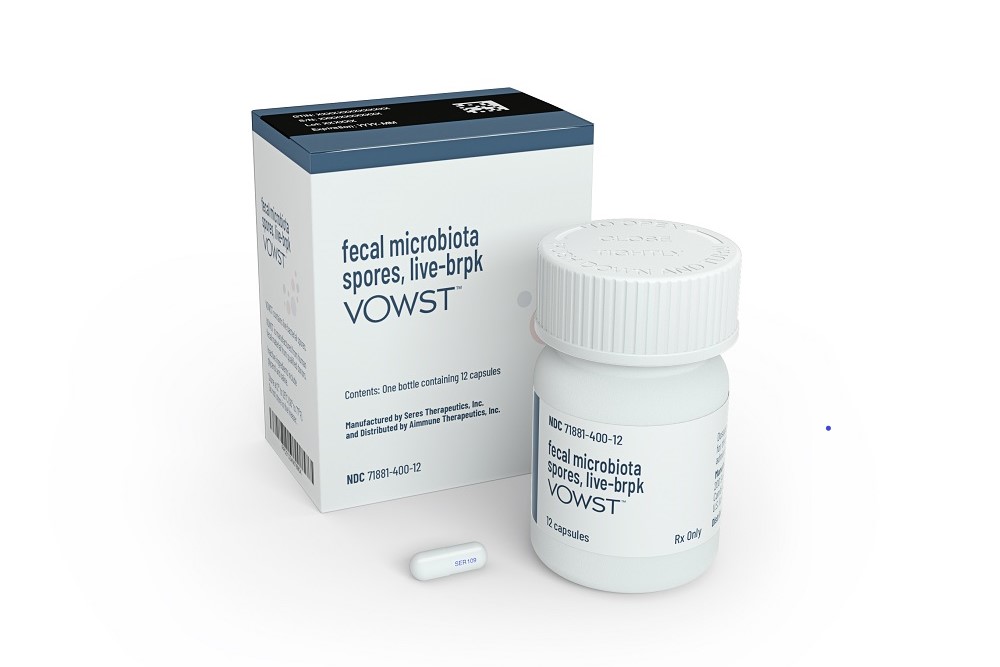
Recurrent Clostridioides difficile infection can be treated with fecal microbiota transplants. FDA approval of Seres Therapeutics’ Vowst makes it the first oral microbiome therapy.

CMS is cracking down on hospital-acquired infections, starting with MRSA and C. difficile.

A 32-year-old woman infected with Clostridium difficile was recently successfully treated with a fecal transplant, an emerging option for people who suffer from different gastrointestinal diseases, commonly caused by antibiotics. The idea is to take donor fecal matter (that has been rinsed and strained) and with either an enema, endoscopy or pill form, replace healthy […]